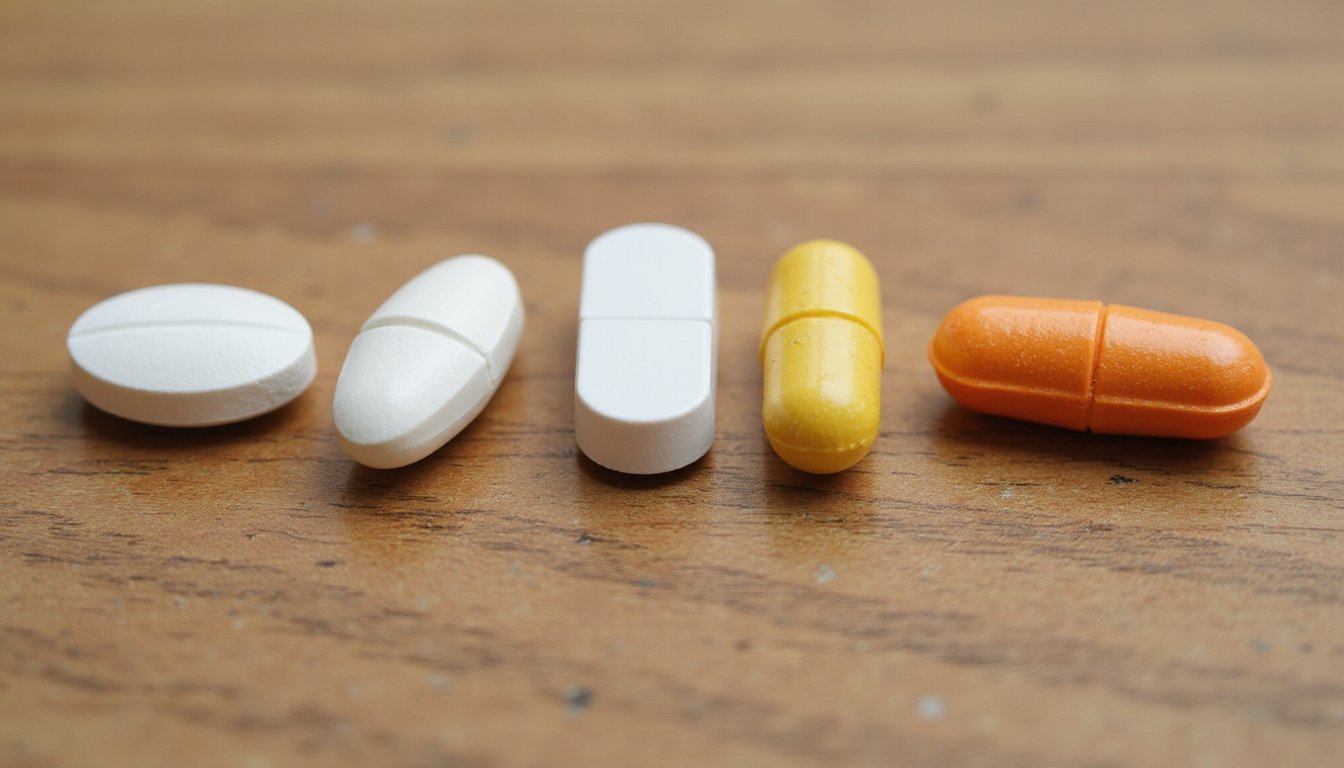
You can identify Norco and Vicodin pills by their unique imprints, colors, and shapes, which vary by manufacturer and strength. Norco tablets typically display codes like IP 109 (5/325), M366 (7.5/325), or M367 (10/325), while Vicodin features markings such as V 3605 (orange, 7.5/300). Most hydrocodone pills are white, oblong, or capsule-shaped, though higher strengths may appear yellow or orange. Below, you’ll find detailed identification guides for each formulation and strength. You can identify Norco and Vicodin pills by their unique imprints, colors, and shapes, which vary by manufacturer and strength, details that matter even as people ask is vicodin still available in the US due to brand discontinuation. Norco tablets typically display codes like IP 109 (5/325), M366 (7.5/325), or M367 (10/325), while Vicodin features markings such as V 3605 (orange, 7.5/300). Most hydrocodone pills are white, oblong, or capsule-shaped, though higher strengths may appear yellow or orange. Below, you’ll find detailed identification guides for each formulation and strength.
Norco vs. Vicodin: Key Differences in Hydrocodone Pills

Both Norco and Vicodin combine hydrocodone with acetaminophen to treat moderate-to-severe pain, but they’re not identical formulations. You’ll find the primary distinction in their acetaminophen content: Norco contains 325 mg while Vicodin contains 300 mg per tablet.
Both medications come in 5 mg, 7.5 mg, and 10 mg hydrocodone strengths. When you’re identifying pills like the ip 109 white pill, understanding these formulation differences matters for safety. These medications work by binding to opioid receptors, hindering the transmission of pain signals between the body and the brain.
The hydrocodone-to-acetaminophen ratios differ slightly between products. Vicodin’s ratios range from 1:60 to 1:30, while Norco’s range from 1:65 to 1:32.5. Both are Schedule II controlled substances with comparable analgesic effects lasting 3-6 hours. Norco’s slightly higher acetaminophen content carries marginally increased liver toxicity risk with repeated dosing. Combining these medications with benzodiazepines or alcohol significantly heightens the risk of overdose.
Norco 5/325 Hydrocodone Markings: IP 109, U01, G035, T257
When you’re identifying a Norco 5/325 tablet, you’ll encounter several distinct manufacturer imprints, with IP 109 appearing on white oblong tablets from specific generic producers. You can differentiate between manufacturers by noting that U01 marks round tablets while G035 and T257 appear on oblong or capsule-shaped versions. Authentic markings should match documented characteristics precisely, including tablet shape, color, and scoring patterns that correspond to verified pharmaceutical databases. These tablets contain hydrocodone and acetaminophen, which work together to treat severe pain by changing how the brain and nervous system respond to pain signals. Due to hydrocodone’s opioid properties, these medications carry risks of addiction and dependence with regular use.
IP 109 Tablet Features
The IP 109 imprint identifies a specific hydrocodone/acetaminophen tablet containing 5 mg of hydrocodone bitartrate and 325 mg of acetaminophen. This ip 109 pill is classified as a Schedule CIII controlled substance and represents a generic formulation of Norco.
You’ll recognize this tablet by its white to off-white color and oblong, capsule-like shape measuring approximately 15 mm. The biconvex design features “IP 109” stamped on one side with a score line on the reverse, allowing you to split the tablet if needed.
The formulation combines an active opioid analgesic with acetaminophen for enhanced pain relief and fever reduction. Hydrocodone works through mechanisms similar to codeine, affecting the central nervous system and smooth muscle. Specifically, hydrocodone binds to receptors in the brain and spinal cord, blocking pain signals and providing relief. Inactive ingredients include colloidal silicon dioxide, magnesium stearate, and microcrystalline cellulose. This tablet meets USP Dissolution Test 1 standards.
Manufacturer Imprint Differences
Although all Norco 5/325 tablets contain identical active ingredients, 5 mg hydrocodone bitartrate and 325 mg acetaminophen, you’ll encounter distinct imprint codes depending on which pharmaceutical company manufactured the product. The ip 109 pill features an oblong white tablet, while the u01 pill appears as a round white configuration. The G035 variant displays an oblong shape with a back ridge, and the t257 pill presents an oblong white-to-peach coloration produced by Ascent Pharmaceuticals, Inc.
Despite visual differences, these tablets maintain dosage equivalency and therapeutic consistency. You should store all variants at room temperature (25°C) in tight, light-resistant containers with child-resistant closures. Each manufacturer receives unique National Drug Codes to distinguish their batches while ensuring standardized potency across all formulations. As a Schedule II controlled substance, all these hydrocodone-acetaminophen formulations carry significant risks, and prolonged use may result in tolerance, dependence, and increased risk of overdose. The medication works by binding to opioid receptors in the brain and spine, providing pain relief that typically lasts 4-6 hours depending on individual metabolism and dosage.
Identifying Authentic Markings
Because counterfeit opioid tablets have become increasingly prevalent in illicit drug markets, you’ll need to verify authentic imprints before assuming any pill contains what its marking suggests. Legitimate Norco 5/325 mg prescription tablets carry manufacturer-specific pill imprints that you can cross-reference through FDA pill identification databases.
Common authentic markings include IP 109 (Amneal Pharmaceuticals), U01 (Aurolife Pharma), G035 (Glenmark Pharmaceuticals), and T257 (Camber Pharmaceuticals). Each appears on white, oval or capsule-shaped tablets with scoring for splitting.
You should examine these characteristics systematically: confirm the imprint matches exactly, verify the white coloring, and check for proper scoring. Don’t rely on color alone, as generic variations exist. When discrepancies arise, consult your pharmacist immediately rather than making assumptions about tablet contents. This verification step is critical because counterfeit pills pressed with fentanyl often carry realistic-looking imprints designed to deceive users. Counterfeit Norco containing stronger opioids like fentanyl can lead to fatal overdoses, making proper identification a potentially life-saving practice.
Norco 7.5/325 Hydrocodone Markings: M366, U02, G036, E112
Anyone handling prescription opioids should understand how to identify Norco 7.5/325 tablets by their manufacturer-specific imprints. Each generic version contains hydrocodone 7.5 mg and acetaminophen 325 mg, but visual markers differ by manufacturer.
You’ll encounter these common imprints:
- M366: White oblong tablet from Mallinckrodt with a score line
- U02: White oblong tablet distinguishable from U01 and U03 variants
- G036: White oblong tablet similar to G035 used for lower strengths
- E112: Pink oblong tablet, making it visually distinct from white generics
- All variants share an oval/capsule shape, often scored for splitting
Verifying these markings helps you confirm prescription authenticity. Since counterfeit opioids pose serious risks, you should cross-reference any unfamiliar tablet against verified pharmaceutical databases before use. Because these tablets contain hydrocodone, they carry a high risk of dependence, addiction, and overdose when not used as prescribed. The score line on tablets like the M366 allows for accurate splitting, but users should always follow their prescribing instructions to avoid dose errors.
Norco 10/325 Hydrocodone Markings: 3601 V, IP110, U03, M367
Moving from the 7.5/325 formulation to the higher-strength Norco 10/325 tablets, you’ll find a different set of manufacturer imprints that distinguish these medications. The 3601 V imprint appears on white, capsule-shaped tablets, while IP110 identifies another generic manufacturer’s version of this dosage.
You’ll recognize the U03 imprint on white, oval tablets containing the 10/325 combination. The M367 code, produced by Mallinckrodt Pharmaceuticals, marks white, capsule-shaped pills with identical active ingredients.
Each Norco 10/325 tablet contains 10 mg hydrocodone bitartrate combined with 325 mg acetaminophen. When taking combination medications containing acetaminophen, exceeding 4 grams daily may cause liver damage. When verifying these medications, you should confirm imprint codes through the FDA Pill Identification Database. Don’t rely solely on color or shape, as generic variations exist. If you’re uncertain about any tablet’s identity, consult your pharmacist for professional verification. If you find hydrocodone that is old, expired, or no longer needed, dispose of it properly by flushing it down the toilet or using a drug take-back program.
Vicodin 5/300, 7.5/300, and 10/300 Hydrocodone Markings
When you’re identifying Vicodin tablets across the 5/300, 7.5/300, and 10/300 formulations, you’ll need to recognize that these specific acetaminophen dosages represent older formulations before the FDA’s 2014 mandate limiting acetaminophen to 325 mg per tablet.
Understanding what does Vicodin look like requires attention to manufacturer-specific imprints. Unlike the ip 115 pill m366 vicodin markings found on some generics, Vicodin-branded tablets display distinct codes:
- V 3605 appears on orange, elliptical pills containing 7.5/325 mg
- WATSON 3202 with red dots indicates 10/325 mg strength
- White, capsule-shaped tablets remain standard across most formulations
- Yellow oval V 3601 identifies 10/325 mg versions
- Multiple generics share strengths but differ in producer imprints
When comparing what does Norco look like pill imprints, you’ll notice similar oblong shapes but different manufacturer codes distinguish each product. If you cannot identify a pill using the imprint code, a pharmacist can look up pills by color, shape, and imprint code while also accessing your medication records for verification.
Hydrocodone Pill Colors and Shapes by Manufacturer
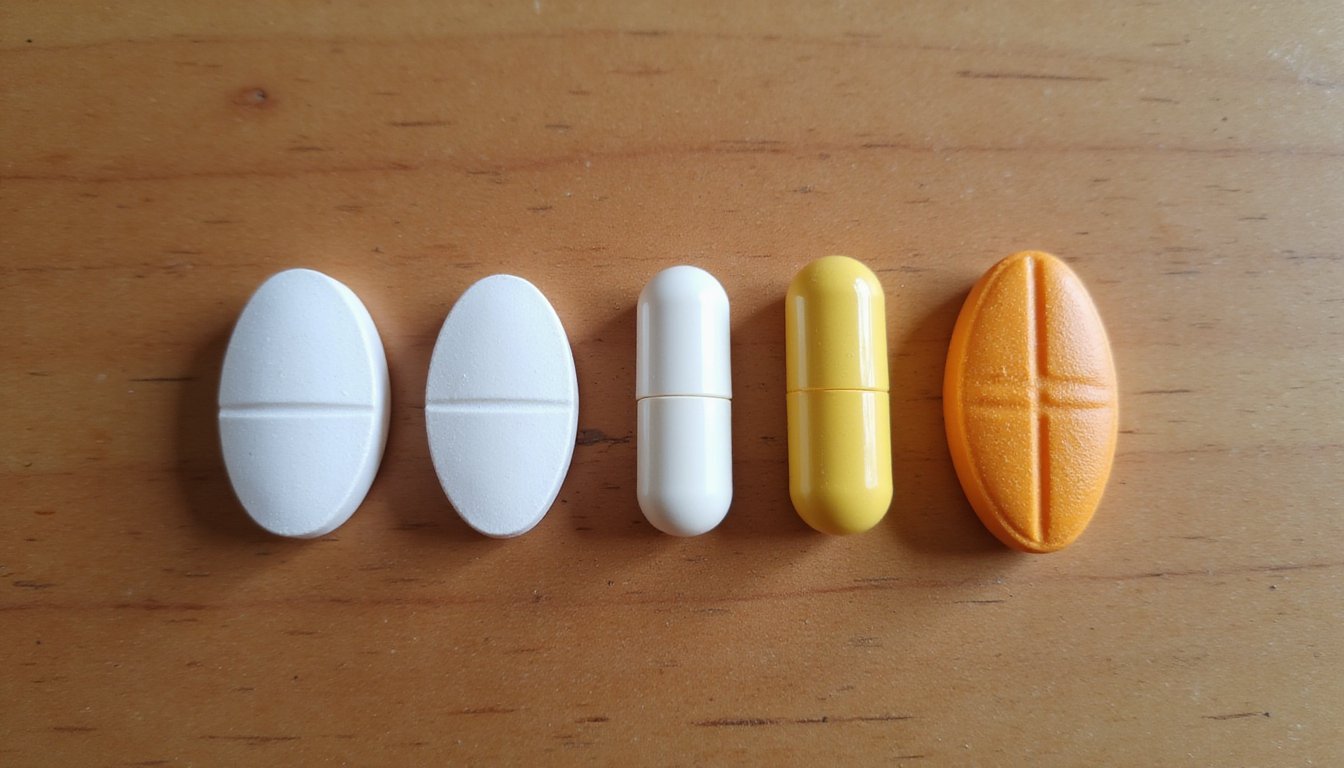
Sorting through hydrocodone tablets by manufacturer requires attention to specific visual markers that distinguish otherwise similar-appearing medications. You’ll find white tablets dominate lower-strength formulations, while yellow pills typically indicate higher-dose Norco 10/325 products. Orange elliptical shapes with V 3605 imprints contain 7.5 mg hydrocodone. Sorting through hydrocodone tablets by manufacturer requires attention to specific visual markers that distinguish otherwise similar-appearing medications, which is why people sometimes ask what is norcom when trying to identify pills by name or appearance. You’ll find white tablets dominate lower-strength formulations, while yellow pills typically indicate higher-dose Norco 10/325 products. Orange elliptical shapes with V 3605 imprints contain 7.5 mg hydrocodone.
When examining manufacturer-specific designs, you’ll notice the g035 white pill features distinct oval shaping, while the t258 pill displays characteristic oblong formatting with clear alphanumeric imprints. The wes301 white pill represents another manufacturer’s approach to standard hydrocodone combinations.
Shape patterns follow predictable trends across suppliers. Oval and capsule-shaped tablets appear most frequently, with scored variants allowing dose splitting when prescribed. Mallinckrodt uses “M” prefixes, Watson employs full name imprints, and other manufacturers utilize letter-number combinations. You should verify any unidentified pill through official FDA databases.
Fake Hydrocodone Pills: Spotting Fentanyl-Laced Counterfeits
You can’t reliably identify fentanyl-laced counterfeit hydrocodone pills by appearance alone, since traffickers replicate exact colors, shapes, and imprint codes like M367 or IP 110. Fentanyl test strips offer a practical screening method, though laboratory analysis using liquid chromatography provides definitive confirmation. The stakes are severe, counterfeit pills containing just milligrams of fentanyl have caused dozens of hospitalizations and deaths, making any pill obtained outside a licensed pharmacy potentially lethal.
Visual Warning Signs
Because counterfeit hydrocodone pills are manufactured to closely replicate authentic Norco and Vicodin tablets, visual inspection alone can’t reliably identify fakes. Counterfeit manufacturers stamp pills with legitimate-looking imprints like “M30” or “E7” to deceive users.
However, you should watch for these potential warning signs:
- Inconsistent coloring or slight shade variations from known authentic versions
- Uneven edges or irregular surface texture
- Powder residue inside packaging or unusual crumbling
- Slight size discrepancies compared to previously verified pills
- Unusual smell or taste differences
Even pills appearing identical to authentic medications may contain lethal fentanyl doses. DEA testing shows 7 out of 10 seized pills now contain potentially fatal amounts. You can’t detect fentanyl contamination without laboratory analysis, visual inspection provides no guarantee of safety.
Fentanyl Testing Methods
Fentanyl test strips offer the only reliable method for detecting fentanyl contamination in suspected counterfeit hydrocodone pills outside a laboratory setting. To test a pill, you’ll need to crush it thoroughly and dissolve at least 10mg in half a teaspoon of water. Insert the wavy end of the strip for 15 seconds, then place it flat for two to five minutes.
Two lines indicate a negative result, even if the second line appears faint. A single line on the left signals fentanyl detection. One line on the right or no lines means you should retest.
You should understand these strips can’t detect all fentanyl analogs, including carfentanil. They also can’t measure fentanyl concentration, only presence. A negative result doesn’t guarantee safety since fentanyl distribution within pills is often uneven.
Counterfeit Pill Dangers
Even with fentanyl test strips in hand, you’re facing a dangerous reality: the counterfeit hydrocodone pill supply has reached unprecedented levels of contamination.
The DEA seized 20 million fake pills laced with fentanyl this year alone, more than the previous two years combined. These counterfeits look identical to legitimate Norco and Vicodin tablets.
Critical facts you need to know:
- Four out of ten fentanyl-containing pills carry a potentially lethal dose
- 26 percent of DEA-tested tablets contained deadly fentanyl levels
- A lethal fentanyl dose equals just 2 milligrams, a few grains of salt
- Fentanyl is 50-100 times more potent than morphine
- Counterfeits are marked to mimic authentic hydrocodone products exactly
In Sacramento, 56 hospitalizations and 15 deaths resulted from counterfeit Norco containing fentanyl instead of hydrocodone. You can’t visually distinguish legitimate pills from deadly counterfeits. In Sacramento, 56 hospitalizations and 15 deaths resulted from counterfeit Norco containing fentanyl instead of hydrocodone. You can’t visually distinguish legitimate pills from deadly counterfeits, which makes questions about dosing, such as difference between 5-325 and 7.5-325, particularly dangerous when pills are obtained outside regulated medical channels.
Your Hydrocodone Pill Doesn’t Match? What to Do Next
When your hydrocodone pill doesn’t match what you’ve received before, you shouldn’t ignore the discrepancy, but you also shouldn’t assume something’s wrong. Generic substitutions frequently cause appearance changes between refills.
Recommended Actions
| Situation | Action | Resource |
|---|---|---|
| Different color or shape | Verify imprint code matches prescription label | Online pill identifier database |
| Unfamiliar imprint code | Contact dispensing pharmacy immediately | Pharmacist consultation |
| No imprint or suspicious appearance | Do not take; seek professional verification | Healthcare provider or pharmacy |
You should compare the imprint code against your prescription documentation first. If codes match but appearance differs, you’ve likely received a different manufacturer’s generic version. When imprint codes don’t match or pills lack identifiable markings, contact your pharmacist before taking the medication.
Frequently Asked Questions
Can I Identify a Hydrocodone Pill if the Imprint Is Partially Rubbed Off?
You can attempt identification, but partial imprints considerably diminish accuracy. You’ll want to note the pill’s color, shape, and any visible characters, then cross-reference using the FDA Pill Identification Database or WebMD’s Pill Identifier. However, you shouldn’t rely on visuals alone due to generic variations. If you can’t confirm the medication’s identity, consult a pharmacist directly. For completely unidentifiable pills, contact Poison Control at 1-800-222-1222 or discard them safely.
Why Do Generic Hydrocodone Pills Have Different Imprints Than Brand-Name Versions?
Generic hydrocodone pills have different imprints because the FDA requires each manufacturer to use unique codes identifying their specific product. While generics contain identical active ingredients and strengths as brand-name versions, companies like Amneal (IP110) or Actavis (2172) must distinguish their pills for traceability. You’ll notice variations in imprints, colors, and shapes between manufacturers, but these differences don’t affect therapeutic equivalence. Always verify imprints through official databases or your pharmacist.
Are Hydrocodone Pill Imprints the Same in Every Country Worldwide?
No, hydrocodone pill imprints aren’t standardized worldwide. You’ll find that each country’s regulatory body establishes its own imprint requirements, meaning a pill manufactured in one nation won’t carry the same codes as those produced elsewhere. The FDA mandates unique imprints for US medications, but no global system exists. If you’re verifying a pill’s identity, you should use country-specific databases and consult a pharmacist, as mismatched imprints may indicate counterfeits.
How Long Have Manufacturers Used Specific Imprint Codes for Hydrocodone Tablets?
You’ll find that manufacturers have used specific imprint codes for hydrocodone tablets since federal regulations took effect in 1995. Before that, only 23 states enforced imprinting laws, and the FDA collected imprint data voluntarily starting in 1986. For example, IP 110 hydrocodone tablets began marketing on February 10, 2010. You can verify a pill’s authenticity by checking its imprint against FDA databases or consulting your pharmacist.
Do Pharmacies Ever Dispense Different-Looking Hydrocodone Pills Than Previously Prescribed?
Yes, pharmacies may dispense different-looking hydrocodone pills than you’ve received before. This occurs when manufacturers change, formulations are updated, or your prescribed product becomes unavailable. Pharmacists can substitute equivalent products after consulting your prescriber, for example, switching from Vicodin to Norco when acetaminophen dosages differ from current formulations. You should always verify any unfamiliar pills with your pharmacist, who can confirm the medication matches your prescription’s intended therapeutic combination.





